|
Understanding Bohr's model requires some knowledge of electromagnetic radiation (or light).īohr's key idea in his model of the atom is that electrons occupy definite orbitals that require the electron to have a specific amount of energy. In 1913, the Danish physicist Niels Bohr proposed a model of the electron cloud of an atom in which electrons orbit the nucleus and were able to produce atomic spectra. These difficulties cast a shadow on the planetary model and indicated that, eventually, it would have to be replaced. Furthermore, Rutherford's model was unable to describe how electrons give off light forming each element's unique atomic spectrum. The Bohr model is a relatively primitive model of the hydrogen atom, compared to the valence shell model. 
If the electron circling the nucleus in an atom loses energy, it would necessarily have to move closer to the nucleus as it loses energy, and would eventually crash into the nucleus. This is, after all, how we produce TV signals. It was already known that when a charged particle (such as an electron) moves in a curved path, it gives off some form of light and loses energy in doing so. Unfortunately, there was a serious flaw in the planetary model. ISBN: 9780754631774.\): Niels Bohr with Albert Einstein at Paul Ehrenfest's home in Leiden (December 1925). Discovering Water: James Watt, Henry Cavendish and the Nineteenth Century Water Controversy. Garden City, NY: Doubleday, 1964.įrayn, M. The Bohrs atomic model although assumes that the energy of the electron depends only on the principle quantum number (n) of the orbit. The electron cloud model has a similar feature of quantized orbits. The Fly in the Cathedral: How a Small Group of Cambridge Scientists Won the Race to Split the Atom. Bohrs model assumes quantized orbits in which the electrons can move without the emission of radiant energy. “ On the Constitution of Atoms and Molecules.” Philosophical Magazine Series 6 26 (July 1913): 1-15.Ĭathcart, B. On the Constitution of Atoms and Molecules. Niels Bohr: His Life and Work as Seen by His Friends and Colleagues. Suspended in Language: Niels Bohr’s Life, Discoveries, and the Century He Shaped. 6.1, “Waves and Electromagnetic Energy.” Electrons change orbits by radiating or absorbing photons.Electrons in stable orbits do not radiate.Orbital angular momentum is quantized hence only certain orbits are possible.Electrons follow circular orbits around a nucleus. 
Explanation of blackbody radiation and atomic spectra.Majority of the mass is found in the nucleus.Conclusions from the gold foil experiment.Electrons are distributed uniformly throughout the atom.He defines the different isotopes of hydrogen. 35 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The nucleus consists of 35 protons (red) and 44 neutrons (blue). He details Bohr’s postulates for the hydrogen atom and discusses how the Planck-Einstein relationship applies to electron transitions. Diagram of the nuclear composition and electron configuration of an atom of bromine-79 (atomic number: 35), the most common isotope of this element. Sadoway talks about the principles of modern chemistry and how that led to the understanding of the structure of the atom. Periodic Table and Table of Constants Lecture Summary Line spectra the Bohr model uses of emission and absorption spectra 6.3, “Atomic Spectra and Models of the Atom.” 6.2, “The Quantization of Energy.”īlackbody radiation the photoelectric effect The electron radioactivity the atomic model Understand Bohr’s quantization condition.Īrchived Lecture Notes #1 (PDF), Sections 1-3.Understand Rutherford’s “nuclear” model. 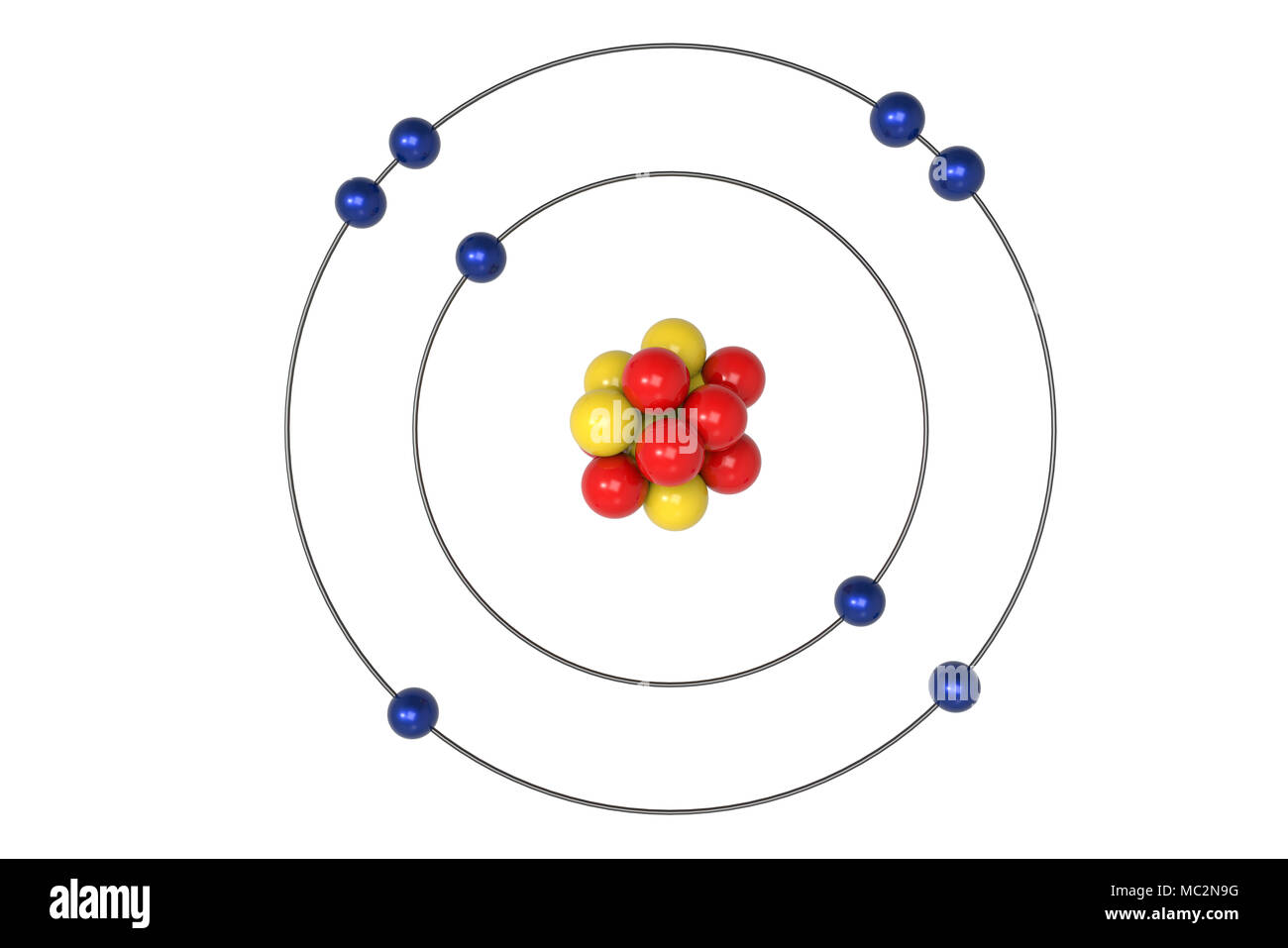
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
